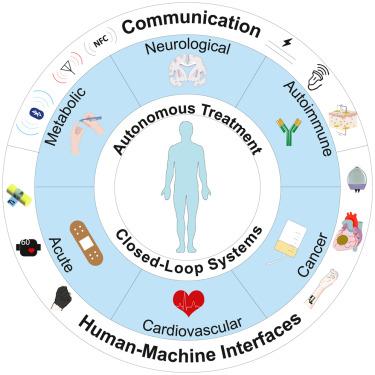
Imagine having a tiny device inside your body that can continuously monitor your health and deliver the right treatment when needed. That's what closed-loop drug delivery systems (CLDDs) can provide, automatically monitoring, adjusting, and administering medication in response to specific signals within the body.
For example, CLDDs can be used to manage chronic medical conditions, such as diabetes, where maintaining precise control over mediation dosage is critical.
While they hold immense promise for improving patient outcomes and treatment adherence, CLDDs have only recently entered clinical use due to the difficulty in integrating the sensing and actuating components of human-machine Interfaces (HMIs).
Researchers at Georgia Tech’s School of Chemical and Biomolecular Engineering have published an article in Device that provides a comprehensive overview of advancements, strengths, and challenges associated with various CLDD approaches.
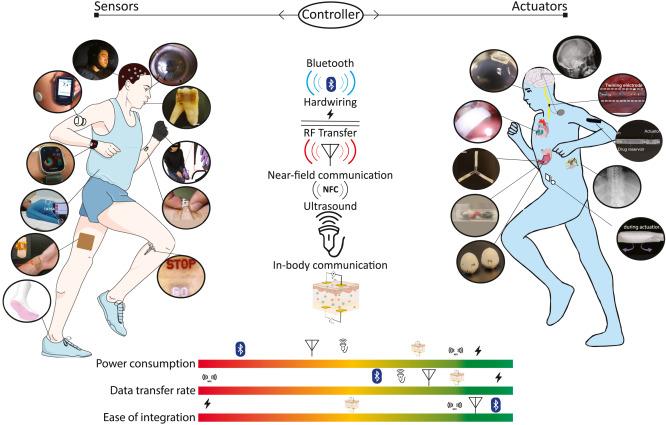
Continuum illustrating state-of-the-art sensors and actuators developed in the past decade.
Examples of devices already in use include insulin pumps, implantable pain pumps, and epilepsy neurostimulators.
In the paper, titled “Communication Protocols Integrating Wearables, Ingestibles, and Implantables for Closed-Loop Therapies,” the researchers explore both passive and active CLDDs.
Passive devices (typically implantable or ingestible) can release drugs over extended periods without active, real-time monitoring, while active CLDDs incorporate real-time monitoring and feedback mechanisms to adjust drug delivery in response to changing circumstances.
“Active closed-loop, drug-delivery systems are poised to usher in a new generation of remote, personalized healthcare driven by human-machine interfaces,” said study co-author Alex Abramson, an assistant professor in Georgia Tech’s School of Chemical and Biomolecular Engineering.
“But to accentuate the shift from passive to active CLDDs, the integration of advanced sensors and actuators is crucial,” added Ramy Ghanim, a PhD student in Abramson’s lab and co-author of the paper.
Sensors in CLDDs continuously monitor specific health parameters in the body (e.g., blood glucose levels for diabetics), and that data is fed to actuators that determine if a specific treatment is needed (such as releasing insulin).
Communication Systems
In the article, the researchers explore various methods for communication transmission in CLDDs, including hardwiring, radio frequency (RF) wireless communication such as Bluetooth, ultrasound, and in-body communication (harnessing the body itself for data transfer through methods like ionic, biochemical, and optical communication). Each method comes with unique advantages and challenges, according to the researchers.
Challenges in developing advanced HMIs include battery size constraints, powering requirements, data transmission rates, and locational dependance.
One big challenge is making sure these devices work no matter where they are inside a patient. Like a cellphone working best near a Wi-Fi router, these devices need to be in the right place to communicate effectively. Sometimes, they move around inside the body, which can be a problem.
The paper explores potential solutions to various challenges, including energy harvesting techniques, wireless powering, and location tracking systems. Ensuring secure data transmission and protection against hacking is also crucial, the researchers noted.
Benefits to Patients
Benefits of CLDDs include simplicity by automating treatment, reducing side effects by delivering medication precisely in a timely manner, and cost-effectiveness by reducing hospitalizations and complications associated with patient non-compliance.
Up to half of all patients requiring frequent and redundant dosages are noncompliant, sometimes missing doses due to complex treatment regimens, according to the researchers. Consequences include decreased quality of life, preventable disease progression, and an estimated annual cost of $528.4 billion in U.S. healthcare expenditure solely due to suboptimal medication therapy.
“Closed-loop drug delivery systems are poised to transform the landscape of chronic illness treatment by enhancing therapeutic release profiles and easing drug administration, thereby improving patients’ quality of life, decreasing medical expenditures, and improving compliance,” Abramson said.
CITATION: Ramy Ghanim, Anika Kaushik, Jihoon Park, and Alex Abramson, “Communication Protocols Integrating Wearables, Ingestibles, and Implantables for Closed-Loop Therapies,” Device, https://www.cell.com/device/fulltext/S2666-9986(23)00144-8, 2023

Professor Alex Abramson

PhD student Ramy Ghanim
Brad Dixon, braddixon@gatech.edu